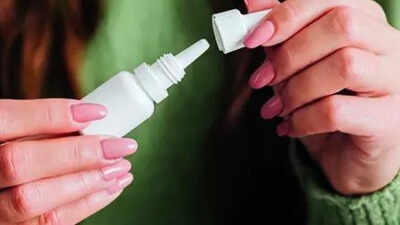
 The Central Drugs Standard Control Organization (CDSCO) in a communication with drug control officers of states and union territories said that “0.3% sodium hyaluronate eye drops” are being manufactured and sold without compulsory permission from the central authority.Officials said the formulation falls under the “new drug” category, which means it must be approved before it can be sold on the market. Without approval, its safety and effectiveness have not been verified.Often used to treat dry eyes and irritation, these eye drops work like artificial tears to keep your eyes moist and reduce discomfort. But experts warn that using unapproved products – even routinely – can lead to complications.For the public, CDSCO’s advice is clear – use eye drops only on the advice of a doctor and buy medicines from reliable sources. A seemingly simple remedy can pose risks if not properly removed.“Using unapproved eye drops to treat dry eye is risky as they can worsen irritation, increase dryness, and increase the risk of infections like conjunctivitis. Since the surface of dry eye is already damaged, untested products may do more harm than good. Even approved eye drops should be discarded one month after opening to avoid contamination,” said Dr. Namrata Sharma, Professor of Ophthalmology, Dr. Rajendra Prasad Centre, AIIMS Center for Ophthalmic Sciences, Delhi.Dr Ashu Agarwal, an ophthalmologist at Delhi’s Apollo Hospitals, said: “Unapproved eye drops are unsafe as their quality, sterility and formulation are not guaranteed. Poor manufacturing may lead to infection and reduced effectiveness due to improper formulation. Patients should use only doctor-prescribed products from reliable companies.”
The Central Drugs Standard Control Organization (CDSCO) in a communication with drug control officers of states and union territories said that “0.3% sodium hyaluronate eye drops” are being manufactured and sold without compulsory permission from the central authority.Officials said the formulation falls under the “new drug” category, which means it must be approved before it can be sold on the market. Without approval, its safety and effectiveness have not been verified.Often used to treat dry eyes and irritation, these eye drops work like artificial tears to keep your eyes moist and reduce discomfort. But experts warn that using unapproved products – even routinely – can lead to complications.For the public, CDSCO’s advice is clear – use eye drops only on the advice of a doctor and buy medicines from reliable sources. A seemingly simple remedy can pose risks if not properly removed.“Using unapproved eye drops to treat dry eye is risky as they can worsen irritation, increase dryness, and increase the risk of infections like conjunctivitis. Since the surface of dry eye is already damaged, untested products may do more harm than good. Even approved eye drops should be discarded one month after opening to avoid contamination,” said Dr. Namrata Sharma, Professor of Ophthalmology, Dr. Rajendra Prasad Centre, AIIMS Center for Ophthalmic Sciences, Delhi.Dr Ashu Agarwal, an ophthalmologist at Delhi’s Apollo Hospitals, said: “Unapproved eye drops are unsafe as their quality, sterility and formulation are not guaranteed. Poor manufacturing may lead to infection and reduced effectiveness due to improper formulation. Patients should use only doctor-prescribed products from reliable companies.”WEB DESK TEAMhttps://articles.thelocalreport.in
Our team of more than 15 experienced writers brings diverse perspectives, deep research, and on-the-ground insights to deliver accurate, timely, and engaging stories. From breaking news to in-depth analysis, they are committed to credibility, clarity, and responsible journalism across every category we cover.


